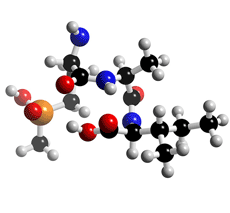
The molecular structure of Phosalacine
Click on the image above to interact with the 3D model of the Phosalacine structure
Phosalacine
Discovery, producing organism and structure (1-3)
Phosalacine was isolated from a fermentation broth of the actinomycete strain KA-338 while screening for L-glutamine competitive antimetabolites. As expected, the compound showed herbicidal activity. The producing strain was classified as a new taxon.
Physical data (1)
White amorphous powder. C15H30N3O6P; mol wt 379.19. Sol. in H2O. Slightly sol. in MeOH. Insol. in EtOH, BuOH, acetone.
Biological activity (1)
1) Antimicrobial activity
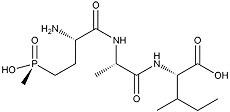
Phosalacine
2) Herbicidal effect on alfalfa (Medicago sativa)
3) Phosalacine almost completely inhibited growth of Bacillus subtilis at a concentration of 0.1 µg/ml, and inhibition was completely overcome upon addition of 10 µg/ml of L-glutamine.
4) Mode of action
Phosalacine decomposed to phosphinothricine
in microbial and plant cells,
and competitively inhibited glutamine
synthetases from B. subtilis and spinach
leaves, with Ki values of 81.1 and 306 µM,
respectively.
References
1. [291] S. Ōmura et al., J. Antibiot., 37, 829–835 (1984)
2. [292] S. Ōmura et al., J. Antibiot., 37, 939–940 (1984)
3. [313] Y. Takahashi et al., J. Gen. Appl. Microbiol., 30, 377–387 (1984)


